November 17, 2017 by Bill Schiffner
fertility treatment, GONAL-f, GONAL-f prefilled pen, Luciano Rossetti, Merck
Pharmacy, Supplier News

DARMSTADT, Germany — Merck has received approval for a new version of GONAL-f (follitropin alfa injection) prefilled pen from the U.S. Food and Drug Administration (FDA). Known as GONAL-f RFF Redi-ject prefilled pen in the U.S. and originally approved by the FDA in 2013, the new version of the pen is easy-to-learn and easy-to-use. “Aspiring
August 14, 2017 by CDR Blog and Chain Drug Review
Kenneth Frazier, Merck, President Donald Trump, protests in Charlottesville
CDR Blog

Amid the shakeout from President Donald Trump’s statements on the violence erupting in protests in Charlottesville, Va., Merck & Co. chairman and CEO Kenneth Frazier said he’s leaving the President’s American Manufacturing Council. Frazier said in a statement posted on Twitter early Monday morning, “I am resigning from the President’s American Manufacturing Council. Our country’s
March 10, 2016 by Chain Drug Review
Kenneth Frazier, Merck, Merck Manufacturing Division, Sanat Chattopadhyay, Willie Deese
Pharmacy, Supplier News

KENILWORTH, N.J. — Sanat Chattopadhyay has been promoted to executive vice president and president of the Merck Manufacturing Division (MMD). Plans call for Chattopadhyay, currently senior vice president of Global Manufacturing Operations (Human Health), to start in the new position on April 1. He succeeds Willie Deese who will retire on June 1 after 12 years
December 18, 2015 by Chain Drug Review
Gardasil, Gardasil 9, HPV, human papillomavirus, Jacques Cholat, Merck, Merck Vaccines
Pharmacy, Supplier News
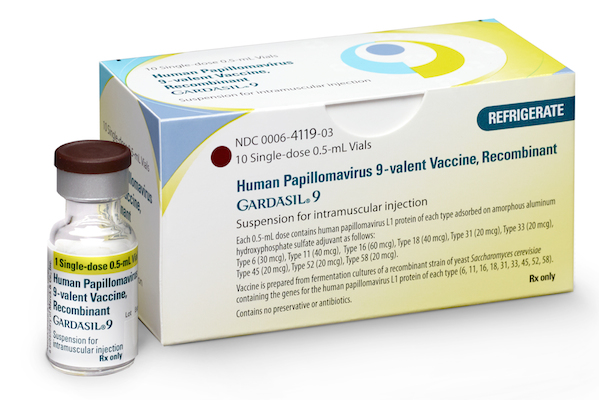
KENILWORTH, N.J. — Merck has received approval from the Food and Drug Administration for an expanded age indication for Gardasil 9, its 9-valent human papillomavirus (HPV) vaccine. The company said the new indication allows Gardasil 9 to include use in males ages 16 to 26 years for the prevention of anal cancer caused by HPV
October 21, 2015 by Chain Drug Review
Marc Greenberger, Market Performance Group, Merck, MPG, Rhonda Johnson
Supplier News

PRINCETON, N.J. — Former Merck executive Rhonda Johnson has joined consulting company Market Performance Group (MPG) as executive vice president and client lead. Johnson was Merck Customer Care’s U.S. Region chief customer officer. “MPG has gained a great resource and leader in Rhonda,” said Marc Greenberger, the company’s managing partner. “She’s a dynamic, seasoned executive
July 16, 2015 by Geoff Walden and Chain Drug Review
Jeffrey Berkowitz, Merck, Pharma and Global Market Access, Walgreens Boots Alliance
Featured Articles, Leading Headlines, Pharmacy, Retail News

DEERFIELD, Ill. — Jeff Berkowitz, executive vice president at Walgreens Boots Alliance and president of Pharma and Global Market Access, has left the company. “With the restructuring of our Pharma and Global Market Access group, Jeff Berkowitz, who has led that function since its formation, has made the decision to pursue other opportunities outside the
April 21, 2015 by Chain Drug Review and Chain Drug Review
Jim Mackey, John Hogan, Johnson & Johnson, Marc Greenberger, Market Performance Group, Merck, MPG, Tim Toohey
Supplier News

PRINCETON JUNCTION, N.J. — Market Performance Group (MPG) announced that founding partner Tim Toohey is retiring and that the company has named two new members to its advisory board. MPG said Tuesday that Toohey is retiring to pursue other business and personal interests but will continue to contribute his knowledge and consulting expertise as a
January 19, 2015 by John Schultz and Chain Drug Review
biosimilar, cancer treatment, Center for Drug Evaluation and Research, FDA, Food and Drug Administration, Generic Pharmaceutical Association, Janet Woodcock, Johnson & Johnson, Merck, Neupogen, Neupogen biosimilar, Ralph Neas, Remicade, rheumatoid arthritis drug, Sandoz, Zarzio
2015, Issue 01-19-2015, News, Pharmacy
WASHINGTON — A Food and Drug Administration panel has recommended that the agency approve what could become the country’s first biosimilar. The panel unanimously urged approval of Sandoz’s filgrastin (Neupogen), a cancer treatment. “This encouraging step forward means that it is very likely now only a question of when, rather than if, filgrastin will be available









