March 31, 2021 by Chain Drug Review
Jon Meacham, NACDS Annual Meeting, Scott Gottlieb
Leading Headlines, Pharmacy

ARLINGTON, Va. — NACDS has announced that Scott Gottlieb, M.D., Commissioner of the U.S. Food and Drug Administration (FDA) from 2017-2019, and Jon Meacham, presidential historian and author, will be the featured speakers at the 2021 NACDS Annual Meeting, which will be held virtually April 26-28. Gottlieb and Meacham will speak during the virtual event’s
March 5, 2019 by Chain Drug Review
FDA, generics, HHS, Scott Gottlieb
Featured Articles, Leading Headlines, Pharmacy

SILVER SPRING, Md. — Food and Drug Administration Commissioner Scott Gottlieb is resigning, effective in about a month. The 46-year-old Gottlieb, who has been commuting weekly to the FDA office here from his home in Connecticut, wants to spend more time with his family, an administration official told The Washington Post. Health and Human Services
February 11, 2019 by Chain Drug Review
Commissioner, Council for Responsible Nutrition (CRN), Food and Drug Administration, president and chief executive officer at CRN, Scott Gottlieb, Steve Mister
Leading Headlines

WASHINGTON — In response to a statement published by Scott Gottlieb, Commissioner, Food and Drug Administration on February 11, the Council for Responsible Nutrition (CRN), the leading trade association for the dietary supplement and functional food industry, issued the following statement: Statement by Steve Mister, president and chief executive officer at CRN: “CRN appreciates the
December 13, 2018 by Chain Drug Review
Council for Responsible Nutrition (CRN), Food & Drug Administration (FDA), Scott Gottlieb
Leading Headlines

WASHINGTON – In response to an announcement made by Scott Gottlieb, commissioner, Food & Drug Administration (FDA) at the Food and Drug Law Institute Enforcement, Litigation and Compliance Conference on December 12, the Council for Responsible Nutrition (CRN), the leading trade association for the dietary supplement and functional food industry, Steve Mister, president and chief
November 9, 2018 by Chain Drug Review
e-cigarettes, Food and Drug Administration, Scott Gottlieb
Leading Headlines, Retail News

SILVER SPRING, Md. — Seeking to curb vaping by minors, the Food and Drug Administration is banning retail sales of most flavored e-cigarettes. FDA Commissioner Scott Gottlieb early this month was on the verge of announcing the prohibition (which will exempt menthol and mint flavors), as well as imposing age-verification requirements for online e-cigarette sales.
June 15, 2018 by Chain Drug Review
Alok Sonig, Buprenorphine, Dr. Reddy's Laboratories, Scott Gottlieb, Suboxone
Pharmacy, Supplier News

HYDERABAD, India and PRINCETON, N.J. — Dr. Reddy’s Laboratories announced today that it has received final approval from the U.S. Food and Drug Administration (USFDA) and is launching buprenorphine and naloxone sublingual film, 2 mg/0.5 mg, 4 mg/1 mg, 8 mg/2 mg, and 12 mg/3 mg, a therapeutic equivalent generic version of Suboxone (buprenorphine and
February 27, 2018 by Chain Drug Review
2017-2018 flu vaccine, Centers for Medicare & Medicaid Services, effectiveness of the flu vaccine, Food and Drug Administration, influenza A H3N2, Scott Gottlieb, World Health Organization
Leading Headlines, Pharmacy, Retail News
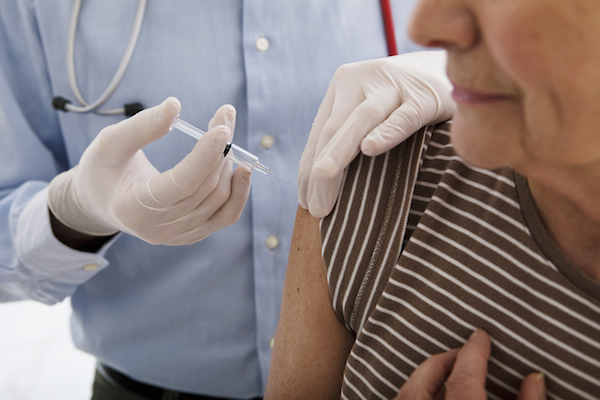
SILVER SPRING, Md. — The Food and Drug Administration has begun efforts to improve flu vaccine effectiveness, according to FDA commissioner Scott Gottlieb. In a statement released yesterday, Gottlieb cited less protection against the influenza A H3N2 virus as a chief reason for the low efficacy of the 2017-2018 flu vaccine. The Centers for Disease
September 25, 2017 by Bill Schiffner
Food and Drug Administration, illegal online pharmacies, International Internet Week of Action, Operation Pangea X, rogue online pharmacies, Scott Gottlieb
Leading Headlines, Pharmacy, Retail News

SILVER SPRINGS, Md. — The Food and Drug Administration has clamped down on hundreds of websites engaged in illegal sales of prescription drugs. The FDA said Monday that, in tandem with international regulatory and law enforcement agencies, actions were taken against more than 500 websites selling potentially dangerous, unapproved versions of prescription medicines — including
May 11, 2017 by Chain Drug Review
Biotechnology Innovation Organization, commissioner of the Food and Drug Administration, FDA commissioner, Food and Drug Administration, Jim Greenwood, Lamar Alexander, Robert Califf, Scott Gottlieb, Stephen Ostroff, Tom Price, U.S. Department of Health and Human Services
Featured Articles, Leading Headlines, Pharmacy, Retail News

WASHINGTON — Dr. Scott Gottlieb has been sworn in as the new commissioner of the Food and Drug Administration. Gottlieb, nominated as FDA commissioner by President Donald Trump in March, took the helm as the FDA’s 23rd commissioner on Thursday after being confirmed by a vote of 57-42 in the Senate late Tuesday. He takes











